A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
A ________,designated by the Greek symbol ψ,describes the wave behavior of an electron in an atom.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following sets of quantum numbers refers to a 4d orbital?
A) n = 2, ![]() = 1,
= 1, ![]() = -1
= -1
B) n = 2, ![]() = 4,
= 4, ![]() = -1
= -1
C) n = 4, ![]() = 2,
= 2, ![]() = -1
= -1
D) n = 4, ![]() = 3,
= 3, ![]() = 0
= 0
E) n = 4, ![]() = 3,
= 3, ![]() = +2
= +2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the wavelength of a photon having a frequency of 54.1 THz? (1 THz = 1015 Hz)
A) 0.180 nm
B) 3.59 × 10-23 nm
C) 1.08 × 10-14 nm
D) 5.55 nm
E) 5.55 × 1015 nm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a proton (mass = 1.673 × 10-27 kg) moving with a velocity of 2.83 × 104 m/s,what is the de Broglie wavelength of the proton (in pm) ?
A) 0.356 pm
B) 3.56 pm
C) 14.0 pm
D) 7.15 pm
E) 28.5 pm
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the energy of a photon of electromagnetic radiation with a frequency of 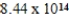 Hz?
Hz?
A) ![]() J
J
B) ![]() J
J
C) ![]() J
J
D) ![]() J
J
E) ![]() J
J
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the Bohr model of the hydrogen atom,the energy necessary to excite an electron from energy level n = 4 to n = 5 is _____ the energy necessary to excite an electron from energy level n = 6 to n = 7.
A) less than
B) greater than
C) equal to
D) either equal to or greater than
E) either less than or equal to
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which type of experiment demonstrates that light has the properties of a particle?
A) nuclear fission
B) electron diffraction
C) light emission from atomic gases
D) mass spectroscopy
E) photoelectric effect
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron in a hydrogen atom,originally in level n = 10,undergoes a transition to a lower level by emitting a photon of wavelength 1739 nm.What is the final level of the electron? (c = 3.00 × 108 m/s,h = 6.626 × 10-34 J·s,RH = 2.179 × 10-18 J)
A) 4
B) 5
C) 10
D) 20
E) 1
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a representation of a 3dxy orbital?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
For which of the following transitions would a hydrogen atom absorb a photon with the longest wavelength?
A) n = 1 to n = 2
B) n = 4 to n = 6
C) n = 5 to n = 4
D) n = 7 to n = 6
E) n = 6 to n = 7
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is INCORRECT?
A) It is not possible to know the exact location of an electron and its exact energy simultaneously.
B) The energies of an atom's electrons are quantized.
C) Quantum numbers define the energy states and the orbitals available to an electron.
D) The behavior of an atom's electrons can be described by circular orbits around a nucleus.
E) Electrons have both wave and particle properties.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 4d orbital has ?
A) 3 planar nodes and 3 spherical nodes.
B) 1 planar node and 1 spherical node.
C) 2 planar nodes and 1 spherical node.
D) 2 planar nodes and 4 spherical nodes.
E) 4 planar nodes and 2 spherical nodes.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following colors of visible light has the longest wavelength?
A) green
B) orange
C) red
D) yellow
E) blue
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the total number of subshells found in the n = 7 shell?
A) 7
B) 49
C) 6
D) 8
E) 9
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
The difference in energy between adjacent energy levels in an atom _____ as n increases.
Correct Answer

verified
decrease.
Correct Answer
verified
Multiple Choice
Which of the following orbital boundary surfaces represent d-orbitals? 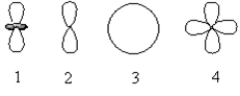
A) 1 only
B) 3 only
C) 2 only
D) 1 and 2
E) 1 and 4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the frequency of gamma ray radiation that has a wavelength of 11.4 pm?
A) 3.80 × 10-20 s-1
B) 1.74 × 10-14 s-1
C) 2.63 × 107 s-1
D) 3.42 × 109 s-1
E) 2.63 × 1019 s-1
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the binding energy of an electron in a photosensitive metal (in kJ/mol) if the longest wavelength of light that can eject electrons from the metal is 299.0 nm?
A) 279 kJ/mol
B) 400 kJ/mol
C) 792 kJ/mol
D) 2.50 × 1019 kJ/mol
E) 4.00 × 10-20 kJ/mol
G) A) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
What is the value of the spin quantum number for an electron in a 3d orbital?
A) 3
B) 2
C) either ![]() or
or ![]()
D) ![]()
E) ![]()
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 65
Related Exams